NovexERP Quarantine & Recall System: Smarter Manufacturing Quality Control
Why Our Quarantine & Recall System is a Game-Changer for Manufacturing Quality Control
In the world of manufacturing and inventory management, few features are as critical—yet often overlooked—as robust quarantine and recall capabilities. Our dedicated Quarantine & Recall module is one of the features we're most proud of, and here's why we built it the way we did.
The Problem: Quality Control Chaos
Most manufacturers have experienced the nightmare scenario: a batch of raw materials arrives with questionable quality, or worse, a finished product already in the field needs to be recalled. Without proper systems in place, these situations devolve into chaos—frantic spreadsheet updates, missed lots, incomplete traceability, and potential compliance violations that can cost companies millions.
Traditional ERPs often treat quality management as an afterthought, burying basic quarantine functions deep within inventory modules or requiring expensive third-party add-ons. We took a different approach.
How We Built It Right
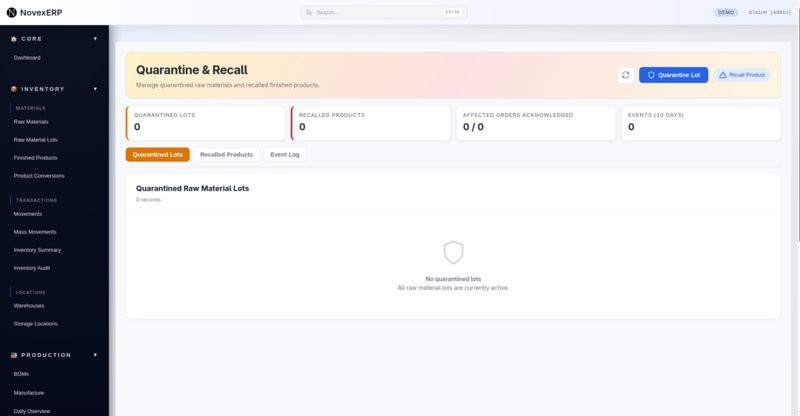
Centralized Command Center
Our Quarantine & Recall interface serves as mission control for all quality-related holds. The dashboard immediately surfaces what matters most:
- Quarantined Lots: Raw material batches on hold, preventing them from entering production
- Recalled Products: Finished goods flagged for customer notification and return
- Affected Orders: Real-time tracking of which orders are impacted
- Event Timeline: A 30-day rolling view of all quarantine and recall activities
This birds-eye view means your quality managers aren't hunting through multiple screens to understand their exposure.
Upstream Prevention
The "Quarantine Lot" function allows your teams to proactively hold suspect raw materials before they contaminate production runs. This upstream intervention is infinitely cheaper than downstream recalls. We've seamlessly integrated this with our raw material lot tracking, ensuring quarantined inventory is automatically excluded from production scheduling and material requirements planning.
Downstream Containment
When finished products need recall, our system tracks which specific orders and customers received affected items. The "Affected Orders Acknowledged" metric ensures follow-up isn't forgotten—a critical compliance requirement in regulated industries like food, pharmaceuticals, and medical devices.
Why Our Customers Love This Feature
Complete Traceability
Our lot tracking creates an unbroken chain from raw material receipt through finished goods shipment. When a recall is initiated, the system can instantly identify every product made from a specific raw material lot, and every customer who received those products. This level of traceability, which might take days with manual systems, happens in seconds.
Regulatory Compliance Made Simple
For industries under FDA, USDA, or ISO scrutiny, audit trails aren't optional. Our Event Log maintains a tamper-proof record of every quarantine decision, every recall initiation, and every order acknowledgment. When regulators come knocking, you're not scrambling to reconstruct events from email threads and handwritten notes.
Financial Protection
Every day a quality issue goes undetected costs money—in wasted materials, scrapped work-in-process, customer compensation, and brand damage. Our clean interface and automated workflows mean quality teams can act faster, contain problems sooner, and minimize financial impact.
Cross-Department Coordination
Quality issues aren't just QA's problem. Production needs to know what materials are off-limits. Sales needs to know which orders are affected. Purchasing needs to know which suppliers are problematic. Our integrated approach means quarantine status flows automatically to every relevant module, eliminating the communication breakdowns that turn small issues into big crises.
Our Design Philosophy
We designed this module with a core principle in mind: quality systems are only effective if people actually use them. That's why we made the interface clean, intuitive, and actionable. No cluttered menus, no nested dialogs, no PhD required to quarantine a lot.
By making quarantine and recall workflows frictionless, we ensure these critical safety nets are actually deployed when needed—not bypassed because the system is too cumbersome.
Our modular architecture means the Quarantine & Recall system isn't a siloed add-on. It's deeply integrated with inventory management, production planning, order fulfillment, and supplier management. This integration is where the real power lies—quarantine a raw material lot, and production schedules automatically adjust. Recall a product, and affected customer orders are instantly flagged.
The Bottom Line
In today's environment of increasing regulatory scrutiny, sophisticated supply chains, and zero-tolerance consumer expectations, manufacturers can't afford quality management systems that are reactive, manual, or fragmented. We built our Quarantine & Recall module to represent a proactive, automated, and integrated approach that turns quality control from a cost center into a competitive advantage.
For manufacturers serious about protecting their brand, satisfying regulators, and sleeping soundly at night knowing they can contain any quality issue within hours instead of days, we've built the tools that make it possible.
That's what NovexERP is all about.